A Simple Urine Test for Detecting Haemolysis
The URICA Rapid Test Kit is a breakthrough lateral flow device developed with the University of Oxford for fast, non-invasive monitoring.
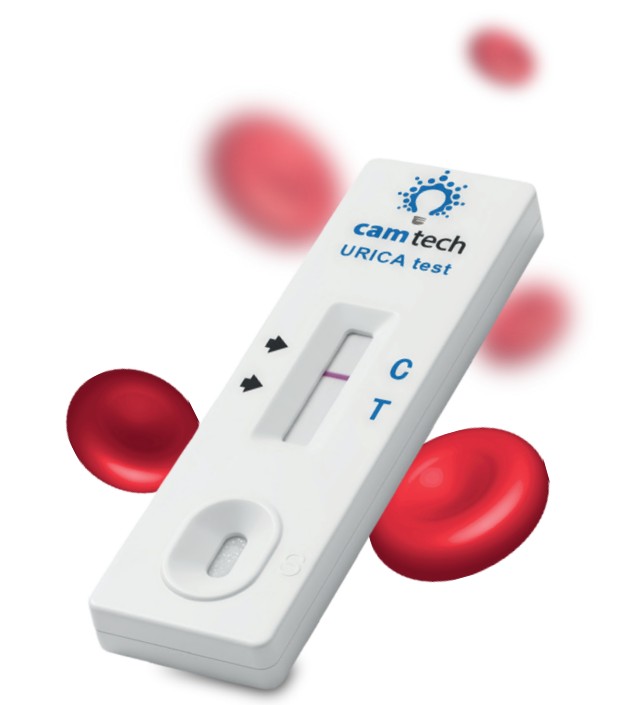
The URICA Rapid Test Kit is a breakthrough lateral flow device developed with the University of Oxford for fast, non-invasive monitoring.

Understanding Red Blood Cell Breakdown
Haemolysis is the process where red blood cells (RBCs) rupture and release their internal contents into the bloodstream. This can occur due to mechanical, chemical, metabolic, infective, or immunological stressors. When this happens inside blood vessels, it is called intravascular haemolysis (IVH).
IVH is a sentinel marker of disease and can lead to Anaemia, Jaundice, Kidney stress, and complications in conditions like Sickle Cell Disease and Malaria.
Traditional diagnostics require blood draws, trained personnel, and lab equipment, leading to delayed turnaround times unsuitable for point-of-care monitoring.

Non-invasive, Fast, and Validated
“The test looks for a unique biomarker in your urine, CA1, which increases when haemolysis occurs.”
CA1 is highly abundant in RBCs and small enough to pass through the kidney filter. It is not normally present in urine, making it a direct and sensitive indicator of intravascular haemolysis.
Rapid detection across clinical and community settings
Early detection of haemolytic crises and home-based monitoring between clinic visits.
Detects haemolysis in newborns with jaundice (ABO incompatibility, sepsis).
Valuable in low-resource settings; more sensitive than plasma-free haemoglobin.
Monitoring for individuals triggered by oxidative drugs or antimalarials.
Helps differentiate between haemolytic and non-haemolytic anaemia in real-time.
Tracks exercise-induced haemolysis for athletes and military training.
CA1 is a direct biomarker of intravascular hemolysis that can be measured routinely in urine using non‑invasive methods under minimal‑laboratory conditions. This urinary CA1 signal has been validated across clinical cohorts using ELISA and western blot and translated into a sensitive lateral‑flow format, demonstrating that CA1 reliably reports ongoing RBC rupture in newborns, inherited anemias, malaria, and other hemolysis‑triggering conditions.
Detection of Intravascular Hemolysis in Newborn Infants Using Urinary Carbonic Anhydrase I Immunoreactivity
The Journal of Applied Laboratory Medicine, Vol 5, Issue 5
View on PubMed → March 2026Urinary carbonic anhydrase 1 excretion is a marker of hemolysis-triggering conditions suitable for point-of-care testing
Blood Glob Hematol . 2026 Mar;2(1)
View on PubMed →
Developed and validated in partnership with Oxford University researchers.